Data
Access data generated by Folding@home projects.
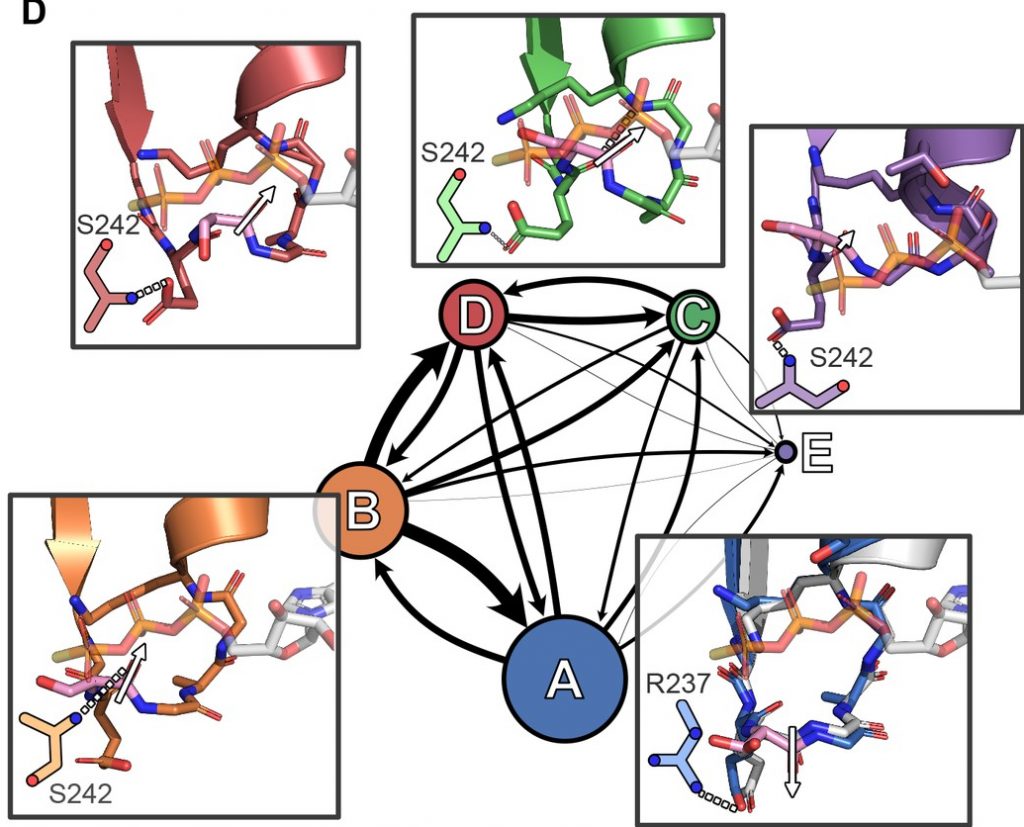
Markov State Models (MSMs) of SARS-CoV-2 proteins
Lab: Bowman Lab, Washington University in St. Louis
In response to the COVID19 pandemic, Folding@home users ran simulations of strategic value for understanding the molecular mechanisms of SARS-CoV-2 and its pathology. For detailed information on this dataset and core highlights, please view our manuscript, “SARS-CoV-2 Simulations Go Exascale to Capture Spike Opening and Reveal Cryptic Pockets Across the Proteome”.
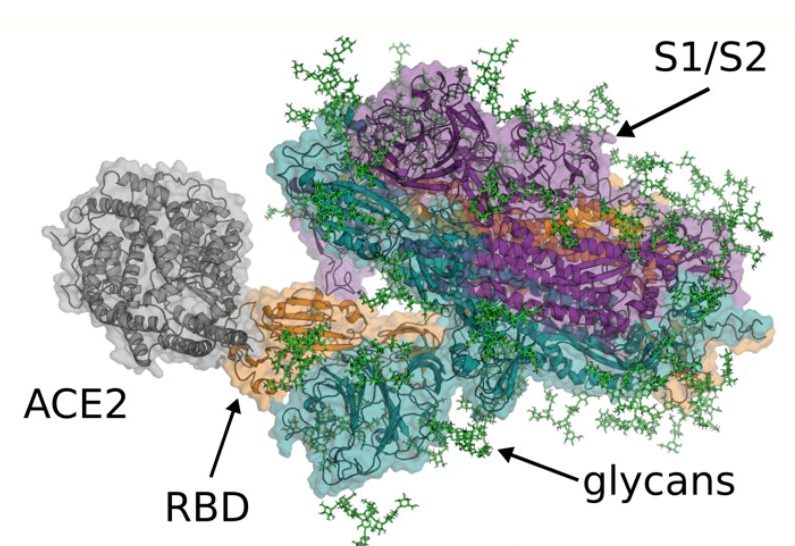
Systems include: SARS-CoV-2: Spike, NSP3, NSP5, NSP7, NSP8, NSP9, NSP10, NSP12, NSP13, NSP14, NSP15, NSP16, Nucleoprotein Receptor Binding Domain, Nucleoprotein Dimerization Domain, SARS-CoV-1: Spike, HCoV-NL63: Spike, Human: ACE2, IL6, IL6-R
SARS-CoV-2 Raw Simulation Data
Lab: Bowman Lab, Washington University in St. Louis
Simulations of SARS-CoV-2 and associated host proteins, with emphasis on discovering druggable cryptic pockets, documented at the MolSSI COVID Hub.
Myosin Motor Markov State Models (MSMs)
Lab: Bowman Lab, Washington University in St. Louis
Myosin motor domains perform an extraordinary diversity of biological functions despite sharing a common mechanochemical cycle. Motors are adapted to their function, in part, by tuning the thermodynamics and kinetics of steps in this cycle. We modeled the distribution of conformations for twelve myosin motor domains by building Markov state models (MSMs) from an unprecedented two milliseconds of all-atom, explicit-solvent molecular dynamics simulations. Porter et al, Conformational distributions of isolated myosin motor domains encode their mechanochemical properties, eLife 2020.