G proteins are essential molecular players in the intricate symphony of cellular signaling. From our vision to our sense of smell, neurotransmission to cell growth, G proteins are at the core of these vital processes. To delve into their activation and unlock the potential for targeted therapies, researchers have embarked on a quest to understand the intricate mechanisms at play. A recent paper takes us on this journey, shedding light on how G proteins of different families are allosterically activated, potentially paving the way for the development of specific treatments for a range of diseases.
G proteins are heterotrimeric complexes, consisting of α, β, and γ subunits. The α subunit, known as Gα, holds the key to their activation (see figure). In their inactive state, Gα proteins have GDP bound between their G domain and H domain. Activation occurs when G protein-coupled receptors (GPCRs) bind to Gα, prompting the exchange of GDP for GTP, and leading to separation from the Gβγ subunit, resulting in the active form of Gα. Despite significant progress in understanding their structure and function, the long-standing challenge has been to fathom how these distant sites communicate, given they are more than 30 Å apart. It is here that molecular dynamics simulations come into play, offering a microscopic view of the intricate process of G protein activation. By running simulations for six different Gα isoforms, researchers aimed to identify commonalities and differences in the allosteric networks that govern their activation.
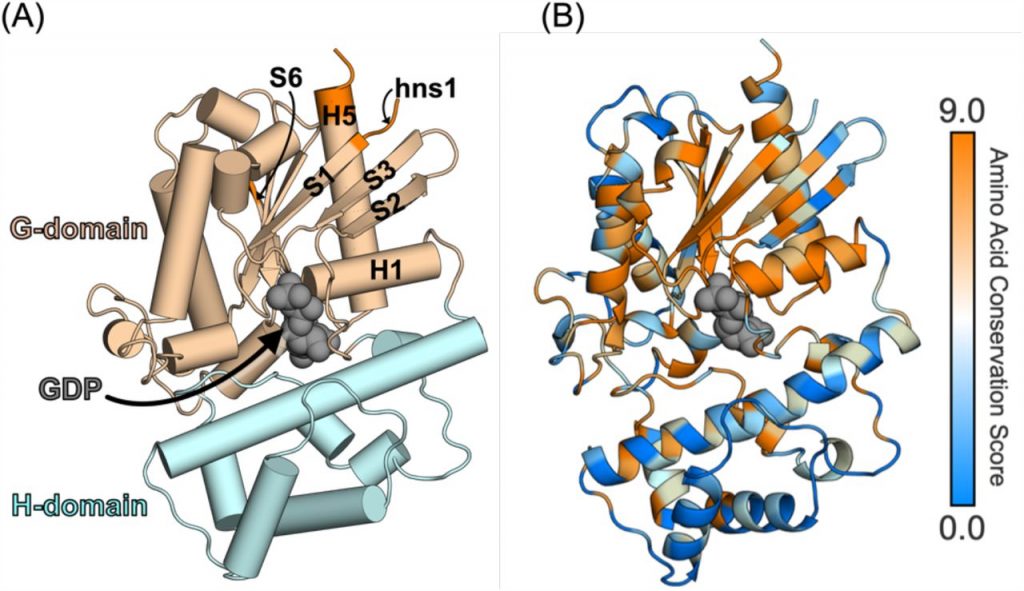
Figure caption: Structure of a Gα subunit and sequence variation across the 16 Gα isoforms. (A) Structure of Gq (PDB: 3ah8) highlighting GDP (grey spheres) bound to the active site, which is between the G domain (light brown) and H domain (cyan). Key secondary structure elements are labeled according to the CGN system. GPCR-binding regions are shown in orange (B) Amino-acid conservation score computed on the Consurf server using the multiple sequence alignment of the 16 Gα isoforms mapped onto the structure of Gq.
The study, which utilized over 210 μs of simulations, focused on Gα isoforms from four different families: Gi/o, Gq/11, G12/13, and Gs. By employing molecular dynamics simulations and building Markov state models (MSM), the researchers could map the conformational space explored by these isoforms. The goal was to uncover the allosteric networks that connect the GPCR-binding and nucleotide-binding sites. In their quest for universality, the research also introduced new methods for assessing the conservation of these allosteric networks. By comparing the isoforms using the Common Gα numbering system, which identifies residues based on their position in secondary structure elements, the study aimed to determine the extent to which G proteins share a universal activation mechanism. This could provide invaluable insights for the development of inhibitors that specifically target different subsets of G proteins, offering hope for tailored treatments for a wide range of diseases.
In conclusion, the recent paper has provided us with a glimpse into the fascinating world of G protein activation. By combining molecular dynamics simulations, Markov state models, and novel methods for assessing allosteric network conservation, researchers are unraveling the intricate mechanisms that underlie the activation of G proteins. As we continue to delve deeper into the differences between G protein isoforms, the potential for developing targeted therapies becomes more promising, offering hope for patients suffering from a range of diseases linked to G protein dysfunction.
