
We’re thrilled to report that the COVID Moonshot has been awarded a $10M grant from the Wellcome Trust on behalf of the World Health Organization (WHO) Access to COVID Tools (ACT) Accelerator program supporting a global effort to fight COVID-19. This funding will enable the Moonshot to rapidly complete its final stages of lead optimization and perform the preclinical studies needed to reach the equivalent of Investigational New Drug (IND) filings to begin clinical trials.
You can find the complete press release here.
The COVID Moonshot aims to discover and develop a patent-free, low-cost oral antiviral drug effective against SARS-CoV-2—a simple pack of shelf-stable pills that would prevent severe COVID-19 disease if taken following exposure, a positive test, or the onset of symptoms—that can be manufactured cheaply and at scale around the world. Oral antivirals will be a key complement to vaccines and antibody therapies in an overall strategy to bring a swift end to the COVID-19 pandemic and help prevent future pandemics. The COVID Moonshot has embraced an open science, crowdsourced approach to achieve this goal, and has already developed an abundant set of highly potent antivirals that are orally bioavailable and effective against multiple SARS-CoV-2 variants of concern. You can read more about how we managed to do this so quickly in our recent Comment in Nature.

For the next stage of the COVID Moonshot discovery effort, we’ve paired with the Drugs for Neglected Diseases Initiative (DNDi), a nonprofit organization with extensive experience in delivering new drugs for global health and ensuring equitable global access to lifesaving medicines. Together with DNDi, the Moonshot will use the Wellcome funding to rapidly complete the final steps of lead optimization in the Discovery phase and rapidly move through the Preclinical phase needed to gain IND approval to begin clinical trials. The Preclinical phase is needed to ensure safety and establish the safe doses that will be used when dosing a new drug candidate into humans for the first time.
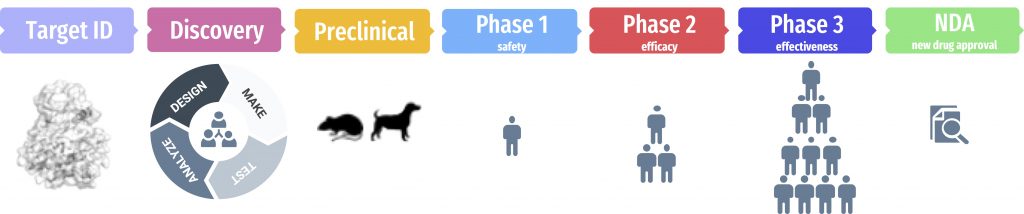
Our objective is to reach clinical trials within a year. Given that much of the world will not receive COVID-19 vaccines until 2024, and that we are continually seeing the emergence of variants (such as Delta, which is proving much more transmissible) that threaten to break through protection afforded by vaccines and therapeutic antibodies, we believe this will still have a significant impact on the course of the pandemic.
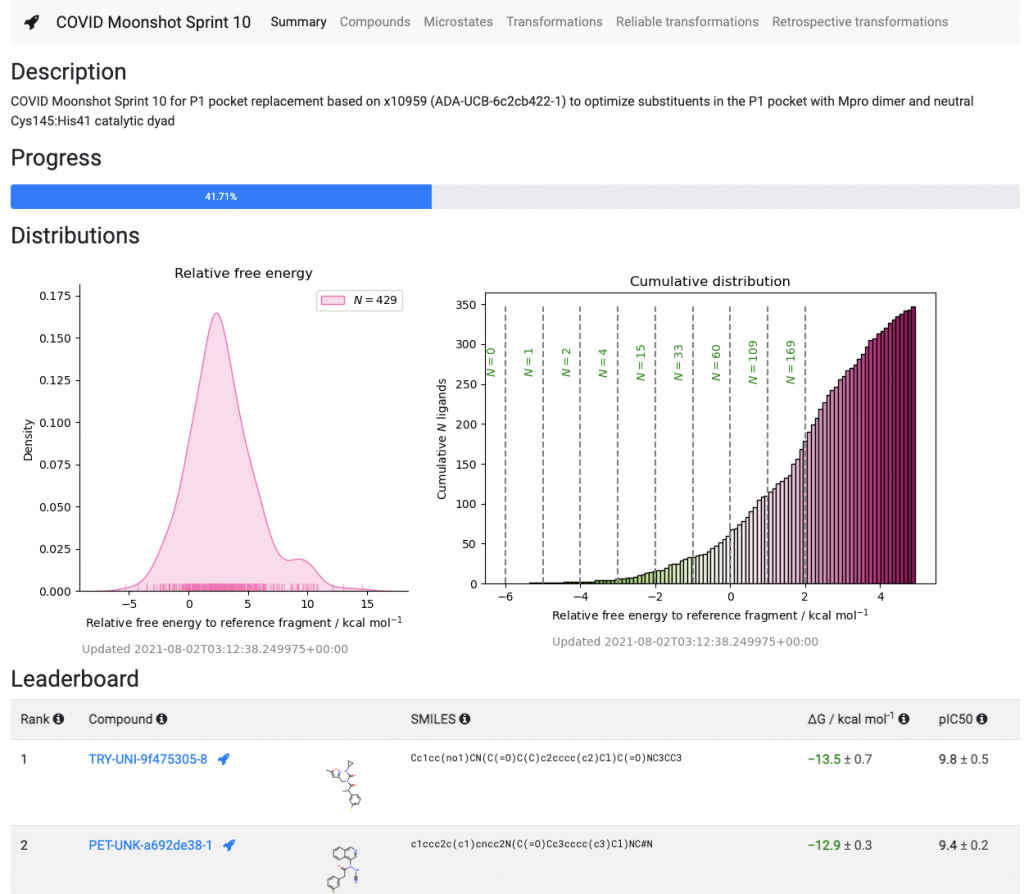
As part of this funding, the Chodera lab at the Memorial Sloan Kettering Cancer Center will receive a small amount of funding to temporarily bring on additional personnel to support the COVID Moonshot sprints on Folding@home. Over the next few months, you’ll see many more COVID Moonshot compound prioritization sprints, following on the recent Sprint 10, which is helping prioritize replacements for part of our compounds that are being rapidly metabolized in rodents. And we’re not stopping with just SARS-CoV-2: We’re hoping that future additional funding, such as the NIH Antiviral Drug Discovery (AViDD) Center program of the new Antiviral Program for Pandemics, which recognizes the need for new oral antivirals like those the Moonshot is developing, will fund the discovery of pan-coronavirus inhibitors to prevent future pandemics.
In the meantime, enormous thanks to everybody that has contributed computer time, support, and funding to Folding@home and the COVID Moonshot program—especially the AWS Disaster Response and Public Datasets teams and the National Science Foundation—we could not have done this without you! Rev up your GPUs and get ready for a few months of intense computing to help us go all the way to clinical trials! 🌙🚀
