Ion channels act as doors for cells: they open and close to let in and out the particles that carry electrical charge, the ions. Ion channels are therefore at the basis of the function of excitable cells such as neurons, cardiac or muscle cells, which all carry small electrical currents that support our movements, thoughts and reflexes.
Viral ion channels can be targeted by ion channel drugs
Viruses also generally use ion channels, although it is not always yet clear how the transport of ions is important for the virus. Viral ion channels, also called viroporins, are thought to be prime possible anti-viral targets.
The story of these drugs starts with Amantadine, in 1966. Amantadine was used for almost 20 years to treat the flu without an understanding of its mechanism of action. 1985 led to a breakthrough, when it was realized that amantadine acted as a blocker for the M2 influenza A virus channel. A little later, it was understood that inhibiting M2 with amantadine reduced the potential of the virus to carry out the early steps of replication. Today, thanks to structural biology, we have a clear picture of how Amantadine blocks the M2 channel, and why certain flu mutations lead to resistance to this drug and its derivatives. Amiloride (sold under the trade name Midamor) is a medicine used to treat, e.g., high blood pressure. It appears to block the Vpu protein from the HIV-1 virus, as well as the Envelope (E) protein from coronavirus, although it is not used to treat any of the infections caused by these viruses. Nobody knows what is the mechanism for this block. This is something we are trying to understand using folding@home!
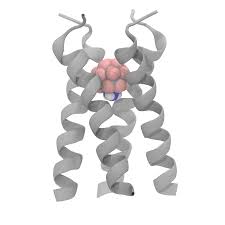
The Envelope protein structure
The Envelope (E) protein is the smallest of the structural proteins that are needed to assemble the virus. Together with the Membrane protein, it appears to be important for the formation of the viral particle in the human cell, because it makes the membrane curve, which is the first step of virus formation.

We don’t yet know what the SARS-CoV-2 Envelope protein looks like at an atomic resolution, but fortunately, we have structures of the SARS-CoV Envelope protein, which is expected to be very similar.
The structures show that the channel assembles as a pentamer, that is, five proteins of the same kind (called monomers) come together to form the pathway along which the ions will be transported. As other ion channels, these proteins are swimming in a see of lipids, which make up the membrane.

Ion channel activity
Recordings of the electrical currents coming from the transport of ions in the E protein of SARS-COV have been done by a technique called patch clamp electrophysiology. These measurements have shown that the protein can transport all kinds of positively charged ions across the membrane, but not negatively charged ones. So far, we do not know how the protein is able to select these ions. This is something that molecular dynamics simulations such as the ones carried out using folding@home can tell us. In the computer, we can apply an electrical stimulus similar to the ones applied in the experiments and see which ions are transported in response.
Ion channel assembly
For the protein to be able to perform its function of transporting ions, it first needs to assemble into a pentamer. In principle, a drug that would prevent it from assembling would do damage to the virus. We would thus like to see the channel assemble, understand how this happens, to later propose strategies to disrupt the process. We hope that with the huge amount of computer power that folding@home provides us, this is possible. The idea to make this happen is to start from a place where the protein pieces (the monomers) are located apart, and launch a bunch simulations. We then only continue the simulations that seem to be going in the direction of the protein assembling, while we stop the ones where the monomers are drifting apart. This strategy is called “adaptive seeding” and is a powerful way to speed up the observation of the processes we are interested in.
Drug block
Once we understand better how the protein works as an ion channel, and how it assembles, we hope that we can understand how amiloride and its derivatives prevent the channel from working properly.
Amiloride is part of the arsenal to treat high blood pressure. It acts by blocking a human ion channel, the epithelial sodium channel ENaC and the sodium–hydrogen antiporter 1 NHE-1, another protein that transports ions across the membranes of cells.

Molecular dynamics simulations can help us understand the mechanisms of drug binding, as described in more details here. With folding@home, we hope we can understand how Amiloride targets the E protein, and to propose ways to make an even better drug therefrom.
E protein role in viral reproduction and vaccine design
There are many strategies for vaccine design. They can be based on weakened or attenuated virus, on viral vectors, on nucleic-acids, on proteins or on virus-like particles.
SARS-CoV and MERS-CoV lacking E-protein have been shown to exhibit significantly reduced viral titers, crippled viral maturation or yield propagation incompetent progeny. Thus viruses lacking the E protein have thus been proposed to serve as a basis for a vaccine. However, optimizing vaccines is a complex process, and understanding basic biology of SARS-CoV-2 can only help further our collective efforts to find Covid-19.
